Compounds and Mixtures.
CK-12 Article (11/29/2018) www.ck12.org/book/CK-12-Physical-Science-For-Middle-School/r1/section/9.2/
Standards
SPS1. Students will investigate our current understanding of the atom.
b. Compare and contrast ionic and covalent bonds in terms of electron movement
SPS2. Students will explore the nature of matter, its classifications, and its system for naming types of matter.
b. Predict formulas for stable binary ionic compounds based on balance of charges.
c. Use IUPAC nomenclature for transition between chemical names and chemical formulas of:
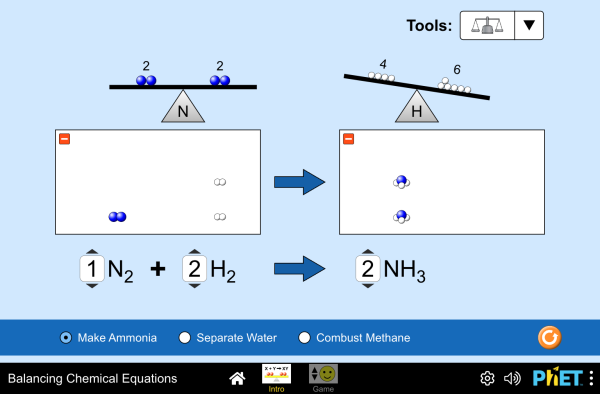
e. Apply the Law of Conservation of Matter by balancing the following types of chemical equations:
a. Describe solutions in terms of
c. Demonstrate that solubility is related to temperature by constructing a solubility curve.
d. Compare and contrast the components and properties of acids and bases.
e. Determine whether common household substances are acidic, basic, or neutral.
b. Compare and contrast ionic and covalent bonds in terms of electron movement
SPS2. Students will explore the nature of matter, its classifications, and its system for naming types of matter.
b. Predict formulas for stable binary ionic compounds based on balance of charges.
c. Use IUPAC nomenclature for transition between chemical names and chemical formulas of:
- binary ionic compounds (containing representative elements).
- binary covalent compounds (i.e. carbon dioxide, carbon tetrachloride).
e. Apply the Law of Conservation of Matter by balancing the following types of chemical equations:
- Synthesis
- Decomposition
- Single Replacement
- Double Replacement
a. Describe solutions in terms of
- solute/solvent
- conductivity
- concentration
c. Demonstrate that solubility is related to temperature by constructing a solubility curve.
d. Compare and contrast the components and properties of acids and bases.
e. Determine whether common household substances are acidic, basic, or neutral.
Resources
|
Naming Compounds
|
|
|
Bonding
|
|
|
Compounds, Mixtures, and Bonding
Balancing Equations
|
|